– Global Expansion of the Phase 2 CRESTONE Study into Canada and Australia
-Case Study of a Patient with Pancreatic Cancer Harboring an NRG1 Fusion and Treated with Seribantumab Under Compassionate Use Highlighted a Confirmed Partial Response and Durable Clinical Benefit
-Strengthened Management Team with the Promotion of Valerie Malyvanh Jansen, MD, PhD, to Chief Medical Officer
NEW YORK, November 12, 2021 — Elevation Oncology, Inc. (Nasdaq: ELEV), a clinical stage biopharmaceutical company focused on the development of precision medicines for patients with genomically defined cancers, today announced financial results for the quarter ended September 30, 2021.
“The third quarter was marked by continued progress on our mission to bring purpose-built medicines to patients with genomically defined cancers,” said Shawn M. Leland, PharmD, RPh, Founder and Chief Executive Officer of Elevation Oncology. “The CRESTONE Study is now open and enrolling patients in the US, Canada, and Australia. Our collaboration with Caris Life Sciences is revealing a series of potential programs targeting genomic fusions. Finally, I am also pleased to welcome Valerie Malyvanh Jansen, MD, PhD, as our first Chief Medical Officer.”
“We are encouraged by the durable clinical benefit seen with seribantumab in the case study of a patient with metastatic pancreatic cancer harboring an NRG1 fusion that was presented at the Australasian Gastro-Intestinal Cancer Trials Group conference,” said Dr. Jansen. “Separately, we expect to complete enrollment of the first 20 patients in Cohort 1 of CRESTONE in mid-2022. We also anticipate presenting initial clinical data at a major medical meeting in mid-2022, which will include results from approximately 10 patients from Cohort 1 with meaningful follow-up who have been treated with seribantumab at 3 grams weekly.”
Recent Business Highlights
Seribantumab
- Case study of a patient with pancreatic cancer treated with seribantumab under a compassionate use program highlighted a confirmed partial response and durable clinical benefit, representing the first clinical data presented on the use of seribantumab in a patient with a tumor harboring an NRG1 fusion. Treatment with seribantumab was included as part of a case series presentation at the Australasian Gastro-Intestinal Trials Group (AGITG) 2021 Annual Scientific Meeting in October. A patient with treatment-refractory metastatic pancreatic cancer who had their tumor genomically profiled through the Cancer Molecular Screening and Therapeutics (MoST) program, was found to harbor an NRG1 fusion, and subsequently received treatment with seribantumab through a compassionate use program provided by Elevation Oncology. As of the data cut-off for the presentation, treatment with seribantumab resulted in durable clinical benefit for over 9 months, an approximately 90% reduction in the cancer biomarker CA19-9, and an ongoing 3 month confirmed partial response per RECIST criteria with a maximum tumor reduction of over 50%.
- Launched additional global clinical trial sites in CRESTONE. The Company recently launched new clinical trial sites in Canada and Australia. CRESTONE is now open and enrolling patients at approximately 30 clinical trial sites across three countries. Through the “just-in-time” clinical site model in partnership with Caris Life Sciences, Tempus, and US Oncology, there are over 400 available sites that can be activated within the CRESTONE study.
Corporate
- Strengthened corporate leadership. During the third quarter, the company announced Valerie Malyvanh Jansen, MD, PhD, as the company’s first Chief Medical Officer. Dr. Jansen was promoted from her prior role as Vice President, Clinical Development.
Upcoming Milestones
- Complete enrollment of the first 20 patients in Cohort 1 of the CRESTONE study in mid-202
- Present initial clinical data from approximately 10 patients from Cohort 1 of the CRESTONE study treated with seribantumab at 3 grams weekly at a major medical meeting in mid-2022
Third Quarter 2021 Financial Results
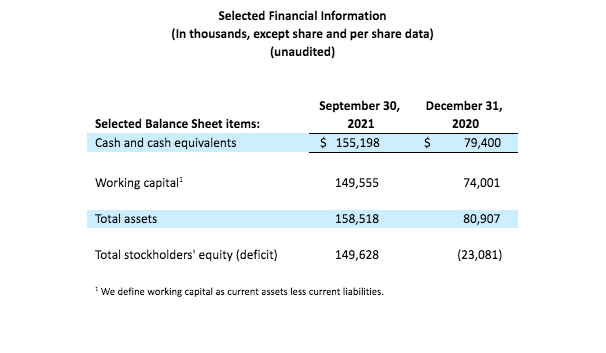
As of September 30, 2021, the Company had cash and cash equivalents totaling $155.2 million, which is expected to fund current operations into the second quarter of 2023.
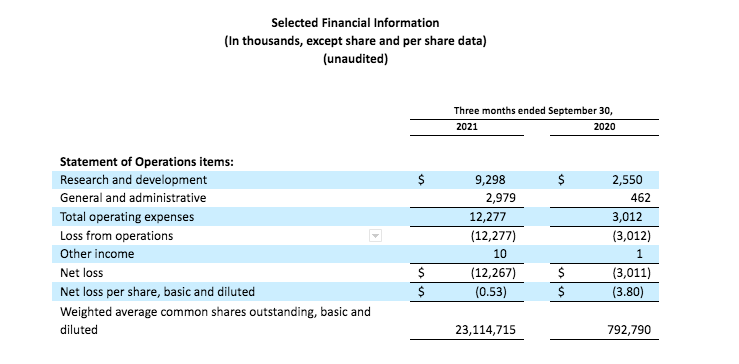
Research and development expenses for the third quarter 2021 were $9.3 million, compared to $2.6 million for the third quarter 2020. The increase in R&D expense was primarily related to an increase in manufacturing and clinical trial expenses associated with the CRESTONE study.
General and administrative expenses for the third quarter 2021 were $3.0 million, compared to $0.5 million for the third quarter 2020. The increase in G&A expense was primarily related to personnel costs, professional services and consulting, and other administrative costs.
Net loss for the third quarter 2021 was $12.3 million, compared to $3.0 million for the third quarter 2020.
About Seribantumab and NRG1 Gene Fusions
Seribantumab is a fully human IgG2 monoclonal antibody that binds to human epidermal growth factor receptor 3 (HER3). HER3 is traditionally activated through binding of its primary ligand, neuregulin-1 (NRG1). The NRG1 gene fusion is a rare genomic alteration that combines NRG1 with another partner protein to create chimeric NRG1 “fusion proteins”. The NRG1 fusion protein is often also able to activate the HER3 pathway, leading to unregulated cell growth and proliferation. Importantly, NRG1 gene fusions are predominantly mutually exclusive with other known genomic driver mutations and are considered a unique oncogenic driver event associated with tumor cell survival.
NRG1 fusions have been identified in a variety of solid tumors, including lung, pancreatic, gallbladder, breast, ovarian, colorectal, neuroendocrine, cholangiocarcinomas, and sarcomas. In preclinical experiments, seribantumab prevented the activation of HER3 signaling in cells that harbor an NRG1 gene fusion and destabilized the entire ERBB family signaling pathway including the activation of HER2, EGFR, and HER4. In addition to extensive nonclinical characterization and testing, seribantumab has been administered to over 800 patients across twelve Phase 1 and 2 studies, both as a monotherapy and in combination with various anti-cancer therapies. Seribantumab is currently being evaluated in the Phase 2 CRESTONE study for patients with solid tumors of any origin that have an NRG1 fusion.
About the CRESTONE Study
Clinical Study of Response to Seribantumab in Tumors with Neuregulin-1 (NRG1) Fusions. CRESTONE is a Phase 2 tumor-agnostic “basket trial” of seribantumab in patients with solid tumors that harbor an NRG1 fusion and have progressed after at least one prior line of standard therapy. The primary objective of the study is to describe the anti-tumor activity and safety of seribantumab as a monotherapy specifically in patients whose solid tumor is uniquely driven by an NRG1 gene fusion. CRESTONE offers a clinical trial opportunity for patients with advanced solid tumors who have not responded or are no longer responding to treatment. Patients are encouraged to talk to their doctor about genomic testing of their tumor. CRESTONE is open and enrolling today in the United States, Australia, and Canada. For more information visit www.NRG1fusion.com.
About Elevation Oncology, Inc.
Elevation Oncology is founded on the belief that every patient living with cancer deserves to know what is driving the growth of their disease and have access to therapeutics that can stop it. We aim to make genomic tests actionable by selectively developing drugs to inhibit the specific alterations that have been identified as drivers of tumor growth. Together with our peers, we work towards a future in which each tumor’s unique genomic test result can be matched with a purpose-built precision medicine to enable an individualized treatment plan for each patient. Our lead candidate, seribantumab, is intended to inhibit tumor growth driven by NRG1 fusions and is currently being evaluated in the Phase 2 CRESTONE study for patients with solid tumors of any origin that have an NRG1 gene fusion. Details on CRESTONE are available at www.NRG1fusion.com. For more information visit www.ElevationOncology.com.
Forward Looking Statements:
This press release contains forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including, but not limited to,
anticipated preclinical and clinical development activities, expected timing of announcements of clinical results, potential benefits of seribantumab and the company’s other future product candidates, potential market opportunities for seribantumab and the company’s other future product candidates, the ability of seribantumab and the company’s other future product candidates to treat their targeted indications, and our expectations about our cash runway. All statements other than statements of historical fact are statements that could be deemed forward-looking statements. These forward-looking statements may be accompanied by such words as “aim,” “anticipate,” “believe,” “could,” “estimate,” “expect,” “forecast,” “goal,” “intend,” “may,” “might,” “plan,” “potential,” “possible,” “will,” “would,” and other words and terms of similar meaning. Although the company believes that the expectations reflected in such forward-looking statements are reasonable, the company cannot guarantee future events, results, actions, levels of activity, performance or achievements, and the timing and results of biotechnology development and potential regulatory approval is inherently uncertain. Forward-looking statements are subject to risks and uncertainties that may cause the company’s actual activities or results to differ significantly from those expressed in any forward-looking statement, including risks and uncertainties related to the company’s ability to advance its product candidates, the timing and results of preclinical studies and clinical trials, approvals and commercialization of product candidates, the receipt and timing of potential regulatory designations, the impact of the COVID-19 pandemic on the Company’s business, the Company’s ability to fund development activities and achieve development goals, the Company’s ability to protect intellectual property, the Company’s ability to establish and maintain collaborations with third parties and other risks and uncertainties described under the heading “Risk Factors” in documents the company files from time to time with the Securities and Exchange Commission. These forward-looking statements speak only as of the date of this press release, and the company undertakes no obligation to revise or update any forward-looking statements to reflect events or circumstances after the date hereof.
Contact:
Argot Partners
212-600-1902
IR@ElevationOncology.com